UFC BIO Products - Free U.S. shipping over $50
Explore UFC Bio’s top-selling sterile solutions and research essentials — including 0.9% sterile saline, USP-grade methylene blue (third-party tested), and a full range of premium buffers, media, and reagents for PCR, DNA sequencing, and other high-precision applications. Proudly made in the USA, every product meets strict quality standards and includes downloadable Certificates of Analysis (CoA) and Safety Data Sheets(SDS).
Free U.S. shipping over $50
-
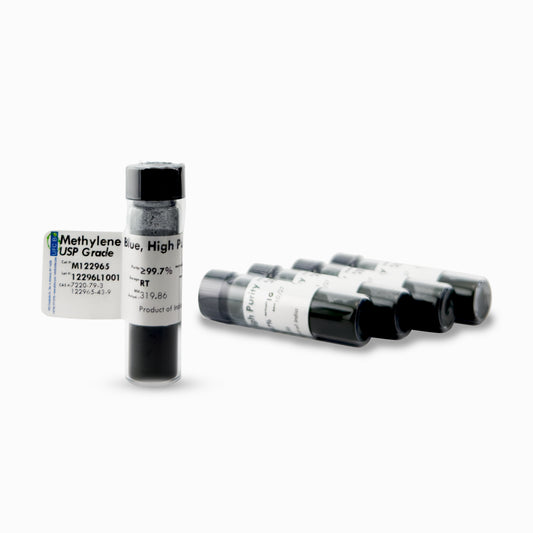
Methylene Blue Powder, USP/Pharma Grade
15 reviews4.93 / 5.0
(15) 15 total reviews
Regular price From $12.50 USDRegular priceUnit price / per -
Methylene Blue USP 1% Solution
30 reviews4.6 / 5.0
(30) 30 total reviews
Regular price $19.50 USDRegular priceUnit price / per -
Sterile Saline Solution, 0.9% - USP Grade - RNase/DNase Free
8 reviews4.88 / 5.0
(8) 8 total reviews
Regular price From $6.00 USDRegular priceUnit price / per -
Sterile Water USP – Ultra Pure Grade
1 review5.0 / 5.0
(1) 1 total reviews
Regular price From $6.00 USDRegular priceUnit price / per -
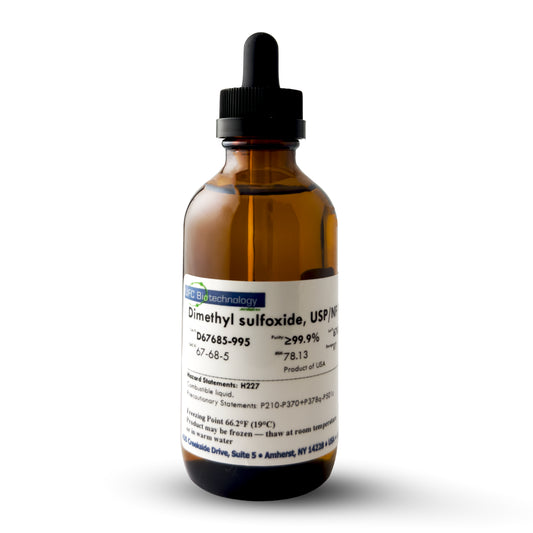
DMSO (Dimethyl Sulfoxide) – 99.9% Pure, USP/NF/ACS Pharma Grade
11 reviews5.0 / 5.0
(11) 11 total reviews
Regular price From $20.00 USDRegular priceUnit price / per -
1X PBS (Phosphate Buffered Saline), 500 mL
No reviewsRegular price $19.99 USDRegular priceUnit price / per